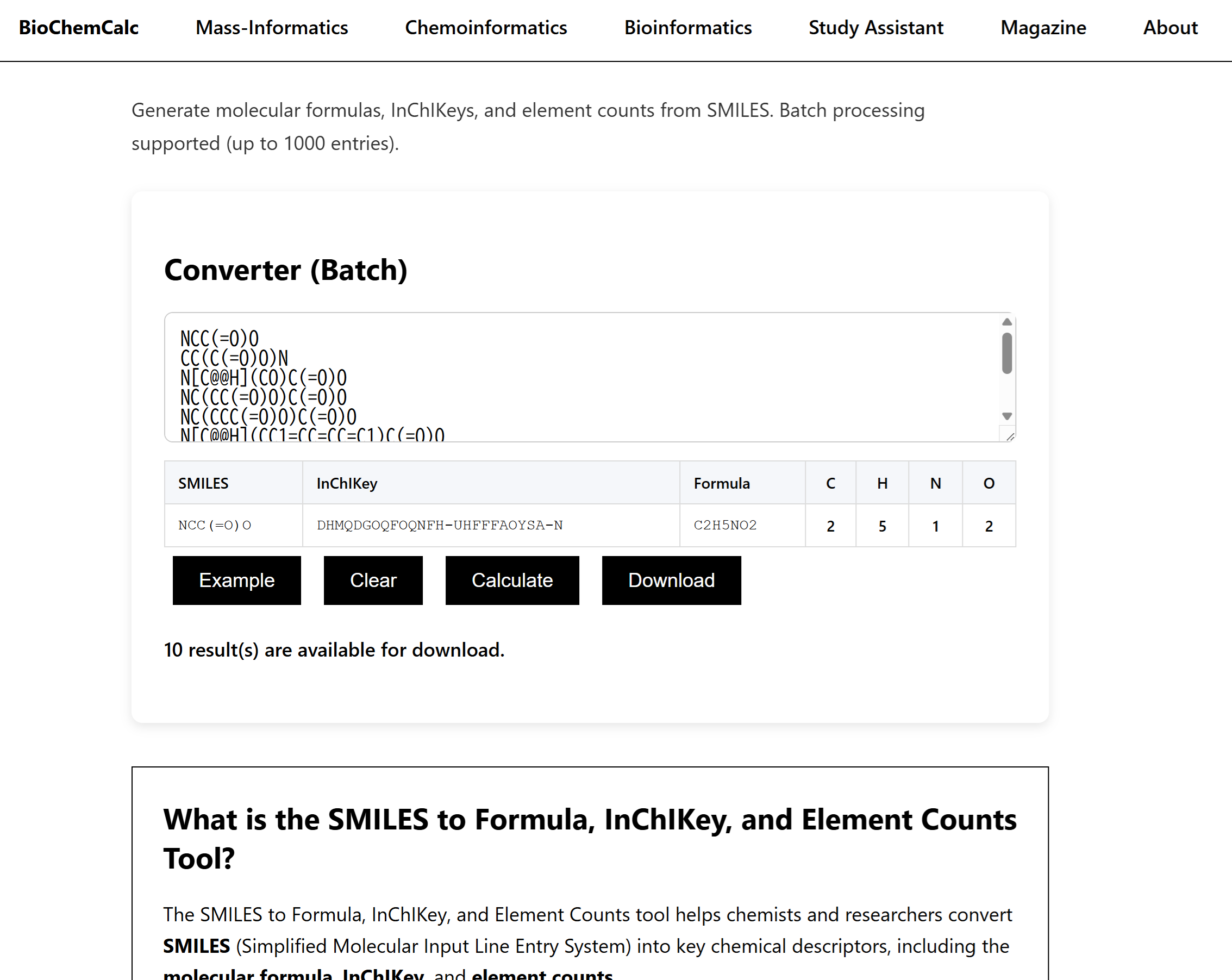
SMILES is a compact and practical way to represent chemical structures.However,a SMILES string by itself is often only a starting point.In many real workflows,researchers need something more structured:a molecular formula,a stable identifier,and a quick summary of elemental composition.
That is why converting SMILES into molecular formula,InChIKey,and element counts can be so useful.This transformation turns a structure string into something easier to compare,filter,validate,and organize.
1.Data Standardization
SMILES is not unique.The same molecule can often be represented in multiple valid ways,which makes raw string comparison unreliable.
By converting SMILES into a molecular formula and an InChIKey,compound lists become much easier to standardize.This is useful when merging datasets,removing duplicates,or preparing clean tables for publications and supplementary materials.
2.Compound Filtering
Element counts make it possible to filter candidate structures using simple chemical rules.Instead of manually inspecting every structure,you can directly ask whether a candidate contains nitrogen,whether sulfur is absent,or whether oxygen count stays within a certain range.
Rules such as N ≥ 1,S = 0,or O ≤ 3 are easy to apply once elemental composition is explicit.This is especially useful in chemoinformatics and mass spectrometry workflows,where a large candidate list often needs to be narrowed down quickly.
3.Consistency Check with MS Data
In analytical workflows,candidate structures should be consistent with observed data.A molecular formula provides one level of validation,but element counts offer a more direct view of composition,especially for heteroatoms such as N,O,P,and S.
For example,a candidate without nitrogen can be excluded when nitrogen is expected.Likewise,unusual elemental compositions can be flagged before deeper interpretation.This makes formula-level validation much easier and more transparent.
4.Data Cleaning
Large compound lists often contain errors.Some SMILES strings may be invalid,some entries may be malformed,and some structures may produce compositions that are clearly inconsistent with expectations.
Element counts provide a simple way to detect these problems.Missing core elements,unexpected atoms,or unrealistic compositions can often be identified immediately.In that sense,this type of conversion is not only informative but also useful for quality control.
5.Batch Processing
Most real projects involve lists rather than single compounds.A useful workflow therefore needs to handle multiple SMILES strings at once,structure the results consistently,and make them easy to export for downstream analysis.
Batch conversion supports exactly this kind of work.It reduces repetitive manual handling and turns a raw list of structures into something ready for spreadsheets,databases,or further computation.
Try the Tool
You can convert SMILES into molecular formulas,InChIKeys,and element counts directly on BioChemCalc here:
https://biochemcalc.com/smi_in_for
Summary
SMILES is useful,but raw SMILES is not always enough.To become practical in research workflows,it often needs to be transformed into structured data that can be standardized,filtered,validated,cleaned,and scaled.
Standardize → Filter → Validate → Clean → Scale.
This is one of the simplest ways to turn chemical structures into usable research data.